I am looking for information as
Researcher

Industry

Patient

“GENETICS CLINIC OF THE FUTURE” SURVEY
Preliminary results from the Genetics Clinic of the Future survey about sharing biobank results with donors
Policies, practices and opinions of BBMRI-ERIC biobanks on sharing biobank results with donors were surveyed by the EU Horizon 2020 funded Genetics Clinic of the Future (GCOF) project. The GCOF survey asked about the current practices and opinions on how this should be performed, with the goal to learn from each other’s experiences and to get background for a possible future guideline by BBMRI-ERIC. The Survey succeeded in reaching at least one biobank in each of the BBMRI-ERIC Member States. Together 72 biobanks replied. The most frequent biobank type was a clinical biobank, and the majority of the respondents identified themselves as biobank directors, heads, managers or similar.
The questionnaire started with questions about national legislation as well as the consent process. The questionnaire then had two sections: the first related to situations where the biobank actively contacts donors offering them (some) results; the second section concerned situations where a donor approaches the biobank with the wish to get (some) results from the biobank. The final part of the questionnaire concerned with opinions about the possible future role of genetics clinics in these processes.
In case of several BBMRI Member States, respondents representing the same Member State had interpreted the national legislation in very different ways. This might reflect the fact that the legislation relating to biobanks is in several countries manifold, maybe scattered and difficult to interpret.
When asked about the policies concerning with the biobank to contact the participant (biobank’s initiative on sharing results), about half of the biobanks informed that they do not have a policy whether or not to share the results with participants. On the other hand, 38 % of the biobanks have a policy to contact participants. A few biobanks have a policy not to share results with participants (Fig. 1).
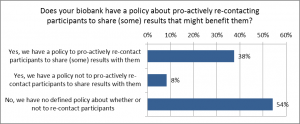
Figure 1. Policies of the biobanks about contacting participants for sharing results. Question no 9 (n=72).
When asked about the policies relating to participants requesting their results from the biobank (participant’s initiative on requesting results), about half of the biobanks replied that they have a policy to return (some) results to the participant. Compared to the previous example, biobanks seemed to have more often a policy to return results if requested (51%) than to actively contact participants for returning results (38%). Of the biobanks 35% informed that they have no policy relating to sharing results with participants who request them. A minority of the biobanks have a policy not to share results if requested (Fig. 2).
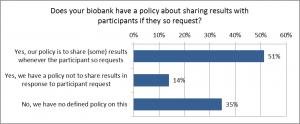
Figure 2. Policies of the biobanks about sharing results with participants following their own initiative to request for results. Question no 22 (n=72).
The majority of the respondents (86 %) answered that they believe that individual results from a biobank could benefit the health of the participants (Fig. 3). These respondents described, for example, that it is important for the participants in order to make lifestyle changes to prevent diseases. The respondents replying that there is no benefit for the participant explained, for instance, that the results come too late to help in health promotion.
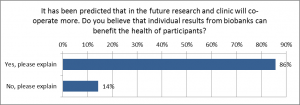
Figure 3. Respondents confidence of the benefit of learning individual biobank results for the health of participants. Question no 34 (n=70).
In this short summary we have presented responses to three separate questions of the Survey. In total, there were over 30 questions and we are now analysing the data. Later this year we will be able to offer a more comprehensive report.
For more information, please contact:
Helena Kääriäinen
Research Professor, MD, PhDNational Institute for Health and Welfare (THL), Finland
helena.kaariainen@thl.fi
tel. +358 29 524 8311
Minna Brunfeldt
Planning Officer, MSc
National Institute for Health and Welfare (THL), Finland
minna.brunfeldt@thl.fi
tel. +358 29 524 7481




